-

Man suspected of stabbing and choking woman in Shelton taken into custody
Police have arrested a man who is suspected of stabbing and choking a woman in Shelton. The victim, a 35-year-old woman, was stabbed at a home on Walnut Tree Hill Road in Shelton on Tuesday night, p... -

Nurses picketing outside Danbury Hospital
Nurses are holding an informal picket outside of Danbury Hospital Thursday amid contract negotiations and said they are asking for support to improve working conditions in the hospital. The Danbury Nu... -

Ten families displaced by fire in Brookfield
Two people have been taken to the hospital to be evaluated and 10 families are displaced after a fire in Brookfield early Thursday morning. Firefighters responded to Boxwood Drive in the Ledgewood Con... -

2 injured, pellet gun fired at police during incident in Hamden
Hamden police are investigating after a man allegedly shot two people and fired at police with a pellet gun in Hamden on Wednesday night. Authorities were called to Spruce Bank Road just before 5 p.m....
-

Clothing drive for families impacted by Avon apartment complex fire
The Avon community is rallying to help dozens of people after a fire at an apartment complex. The flames broke out earlier this week. Now, folks are collecting clothes and gift cards to help families get back up on their feet. With bags full of clothes, people arrived as the doors opened for the first of three days of a…
-

CT Senate passes wide-ranging bill to regulate AI. But its fate remains uncertain
The Connecticut Senate pressed ahead Wednesday with one of the first major legislative proposals in the U.S. to rein in bias in artificial intelligence decision-making and protect people from harm, including manufactured videos or deepfakes. The vote was held despite concerns the bill might stifle innovation, become a burden for small businesses an... -

Man with knife threatens drivers in Bridgeport: police
Police are investigating after a man allegedly used a knife to damage passing vehicles while standing in the middle of the street in Bridgeport. Officers were called to the area of Harral and Lexington avenues just after 6:30 p.m. on Wednesday. A man reportedly made threats while armed with a knife in plain view, according to police. Emergency pers... -

Injuries reported in crash involving stolen vehicle on Route 83 in Vernon
A portion of Route 83 is closed in Vernon after a car crash involving a stolen vehicle on Wednesday night. Vernon police said part of Route 83 North is closed in the area of Cumberland Farms. Drivers are being asked to avoid the area. Police said minor injuries are being reported. No additional information was immediately available.
-

Arrest warrant details events before deadly fire in Wallingford
A prior arrest warrant is shedding light on the events leading up to a deadly house fire in Wallingford. Police say a woman and a 9-month old had passed away before a fire on Geneva Avenue last week. Authorities say Justin Varnado later died. Officers call it a suspicious death investigation. NBC Connecticut has obtained a previous arrest warrant f... -

House approves expanded paid sick leave to cover all workers
House Democrats passed an expansion to the state’s paid sick leave law as part of a busy day. The vote came hours after more than 500 nonprofit service provider employees and clients rallied outside the capitol in support of more funding. Democrats approved the paid sick leave expansion with an 88-61 vote, sending the bill to the Senate. “I think&#... -

Nonprofits rally at Capitol for more funding
Nonprofit service providers rallied at the Capitol Wednesday to push for more funding next fiscal year. Democrats in the legislature say nonprofits are among their priorities, but they’re still negotiating how to spend leftover funds next year. Nonprofits say they’ve been underfunded for years and need additional funding to continue serving their c... -

Leaders visit Portland's Jobs Pond, aim to secure federal funding to mitigate flooding
State and local leaders are trying to do more to help the families who live along Jobs Pond in Portland and are experiencing severe flooding. Environmentalists say the flooding is due to heavy rainfall and because Jobs Pond doesn’t have an outlet to properly drain the water — meaning, when water gets in, it gets stuck. “... -

76-year-old Hartford man wants his diploma decades after expulsion from school
A 76-year-old man from Hartford said he wants his high school diploma 56 years after he was set to graduate. Gerald W. Fothergill, or Mike as many know him, attended Hartford Public High School in the 1960s. In his senior year, he said he was expelled by the school board for walking out of school the day after the assassination…
-

Person injured after jumping out of window during fire in West Hartford
A person was taken to the hospital after jumping from a second floor window during a fire at a West Hartford home. Firefighters responded to a multi-family home on Elmhurst Street for a reported kitchen fire. Responding crews saw heavy smoke coming from the second floor. Shortly after, crews found a resident outside with injuries. They learned that... -
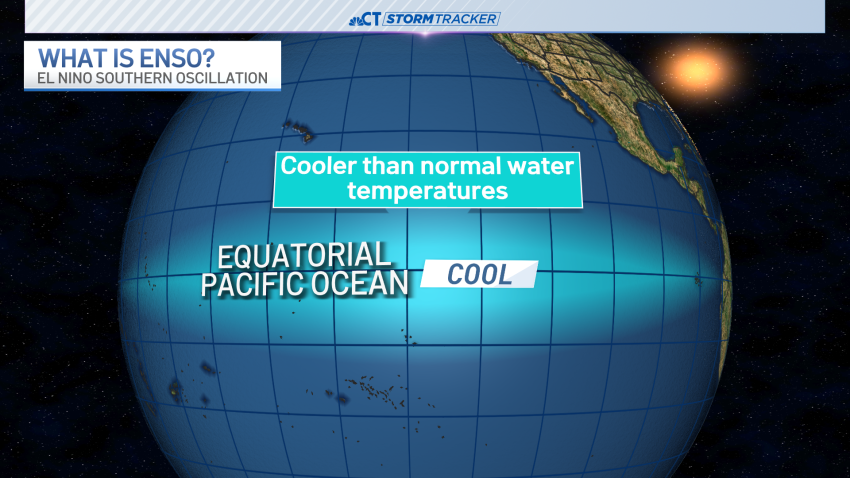
Why hurricane season could be busier than normal this year
Hurricane season will officially begin on June 1 in the Atlantic and May 15 in the eastern Pacific. Looking back, 2023 was a slightly above average season with 20 named storms, seven hurricanes and three major hurricanes. And this year looks like it could be busier, too. A number of universities and other organizations released their Atlantic hurri... -

Motorcyclist arrested for speeding and driving recklessly in Manchester: police
A motorcyclist has been arrested after he was caught speeding and driving recklessly in Manchester on Tuesday. State police said they saw a motorcyclist weaving through traffic, tailgating and passing vehicles unsafely. The driver got onto Interstate 84 West and continued onto I-384, where he was clocked driving over 100 miles per hour. A trooper a... -

Big Brothers Big Sisters honors brotherly bond and recognizes volunteer
Big Brothers Big Sisters of Connecticut is honoring some of their volunteers at the Bigs of the Year Awards. The celebration is at Mark Twain House and Museum in Hartford Wednesday night. Joey Redmond, of Higganum, is named Big Brother of the Year, and said it is only due to the special relationship born from the program. With banter and…...




