- While most people are confident in Covid vaccines, in general, just 46% of survey respondents said they were at least somewhat confident in the J&J shot
- That compares with 69% who said the same for both the Pfizer and Moderna vaccines.
- One in 5 unvaccinated respondents said the pause on J&J's vaccine changed their mind about getting the vaccine in some way.
Less than half of Americans are confident in the safety of the Johnson & Johnson Covid-19 vaccine after it was temporarily halted in the U.S. following reports of a rare blood clotting issue in some recipients, according to a new survey by the Kaiser Family Foundation.
While most people are confident in Covid vaccines, in general, just 46% of survey respondents said they were at least somewhat confident in the J&J shot, compared with 69% who said the same for both the Pfizer and Moderna vaccines. Kaiser surveyed 2,097 randomly selected adults ages 18 and older from April 15 through April 29 for the study published Wednesday.
The Food and Drug Administration and Centers for Disease Control and Prevention on April 13 asked states to temporarily halt using J&J's vaccine "out of an abundance of caution" following reports of rare blood clots in six women. A CDC panel recommended the U.S. resume using the vaccine 10 days later, saying the benefits outweigh the risks.
Get top local stories in Connecticut delivered to you every morning. Sign up for NBC Connecticut's News Headlines newsletter.
The J&J news appears to have changed some minds about getting a shot.
One in 5 unvaccinated respondents said the news altered their view about getting the vaccine in some way, though the specific reactions varied, with 7% saying it made them less likely to want any of the three Covid vaccines, Kaiser found. Another 9% said it made them less likely to want the J&J vaccine but that it didn't change their minds about the Pfizer or Moderna shots.
Money Report
Nevertheless, the share of respondents who say they have received a shot increased significantly from last month's survey, jumping from 32% to 56%. That figure mirrors data from the CDC, which reports that about the same share of U.S. adults have received one dose or more.
"The news was widespread, and it certainly affected confidence in J&J, but it's not clear that it had a big impact on whether people actually got vaccinated or not," said Dr. Mollyann Brodie, executive director of the foundation's public opinion and survey research program. "It confirmed for people who were concerned about side effects that there were side effects, but we know that the immediate effect — at least in terms of what people told us — is very minor in terms of demand."
Women were more likely than men to say the J&J news changed their minds about getting vaccinated, the Kaiser survey showed, particularly Hispanic women, 18% of whom said it made them less likely to want a vaccine at all.

Johnson & Johnson, in a statement to CNBC, said it was collaborating with health authorities around the world to ensure rare blood clots can be identified early and treated effectively.
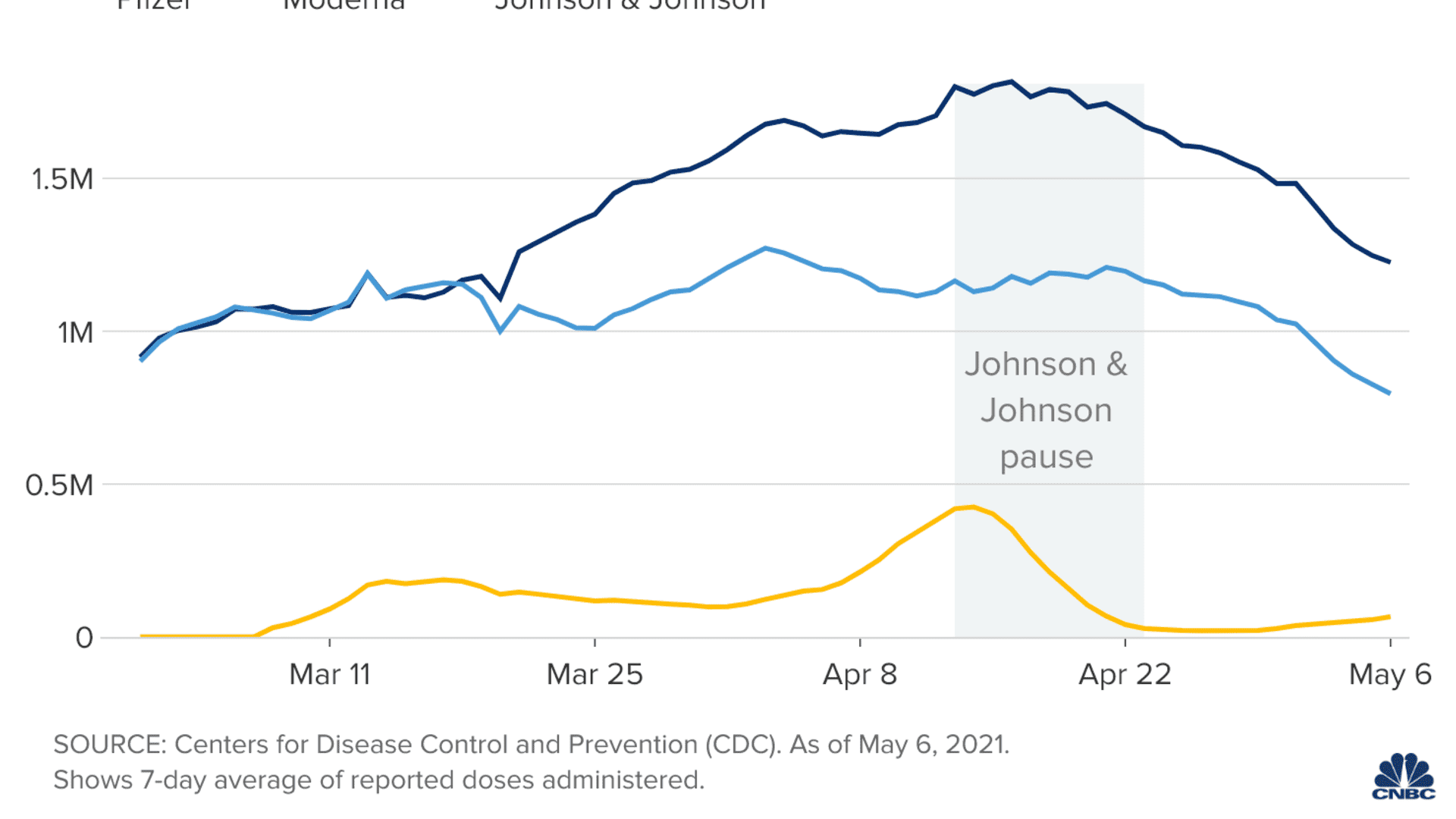
The timing of the Johnson & Johnson pause coincides with an overall slowdown in U.S. vaccinations. The country reported an average of 2.1 million vaccinations per day over the past week, CDC data shows, down from a peak of 3.4 million on April 13.
The fact that the nationwide decline in daily shots occurred during the halt is more of a coincidence than a direct effect, said Dr. Rupali Limaye, a faculty member at the Johns Hopkins Bloomberg School of Public Health. Limaye studies vaccine decision-making and has been working with state health departments during the vaccine rollout.
While the pause on J&J and any resulting reluctance contributes to the decline somewhat, Limaye said the bigger factor is that the country has reached the point where most Americans who want a vaccine have gotten one.
"I'm hearing from states that things are not just slowing generally because of J&J, but slowing because we've essentially been able to satisfy demand," she said.
The Kaiser Foundation survey data backs this up. Respondents who said they were most eager for a shot — those already vaccinated or who want it as soon as possible — increased only slightly from the last survey in March, from 61% to 64%. The share who want to "wait and see" before getting vaccinated, which had been shrinking in size, remained about the same.
"We're at a stage in the vaccination effort where all the eager folks are already vaccinated or in the process," Brodie said. "We're now turning to the people who are hesitant, with strategies needed to reach a lot of different people."






